- Daily & Weekly newsletters
- Buy & download The Bulletin
- Comment on our articles
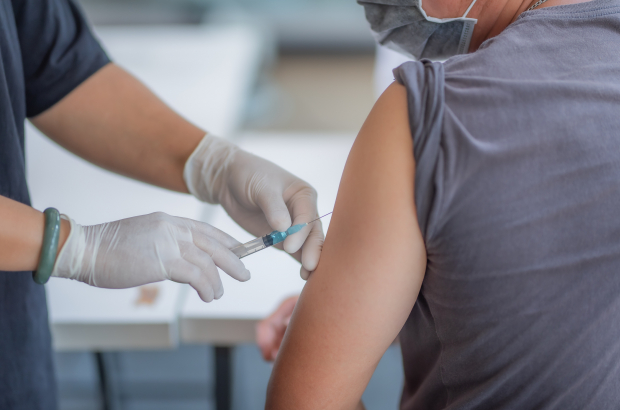
Coronavirus vaccine to be tested on volunteers in Belgium
Janssen Pharmaceutica, headquartered in Beerse, Antwerp province, will start testing a vaccine against Covid-19 next month. While parent company Johnson & Johnson will test the vaccine on volunteers in the US, Janssen will do the same in Flanders.
If everything runs smoothly with the clinical trial, a vaccine could be available as early as the first half of 2021. The clinical trial follows months of lab research in the US.
The human clinical trial is randomised and placebo-controlled, meaning that a randomly chosen group of the more than 1,000 adults taking part in the testing in Flanders will get a placebo rather than the actual vaccine. The trial is also double-blind, meaning that neither the researchers nor the volunteers will know who is getting the placebo and who the vaccine.
Most of the volunteers will be between the ages of 18 and 55, and a smaller number will be over 65. The study will evaluate the immune response and safety of the vaccine.
“As the Company progresses the clinical development of its investigational SARS-CoV-2 vaccine, it continues to increase manufacturing capacity and is in active discussions with global partners to ensure worldwide access,” says Johnson & Johnson in a statement. “The company committed to the goal of supplying more than one billion doses globally through the course of 2021, provided the vaccine is a safe and effective.”
At the beginning of the year, Johnson & Johnson began testing 12 candidate vaccines. “The progress and outstanding results in animals was such that we had a final candidate vaccine much sooner than we had expected,” Johan Van Hoof, head of vaccines at Janssen Pharmaceutica, told VRT. “It’s still in development, so we cannot guarantee anything right now. But on the basis of the success in animals that we had expected, we also expect to have a vaccine in the first half of next year.”
Anyone interested in volunteering for the clinical trial can sign up here.
Photo ©Udom Pinyo/iStock/Getty Images Plus